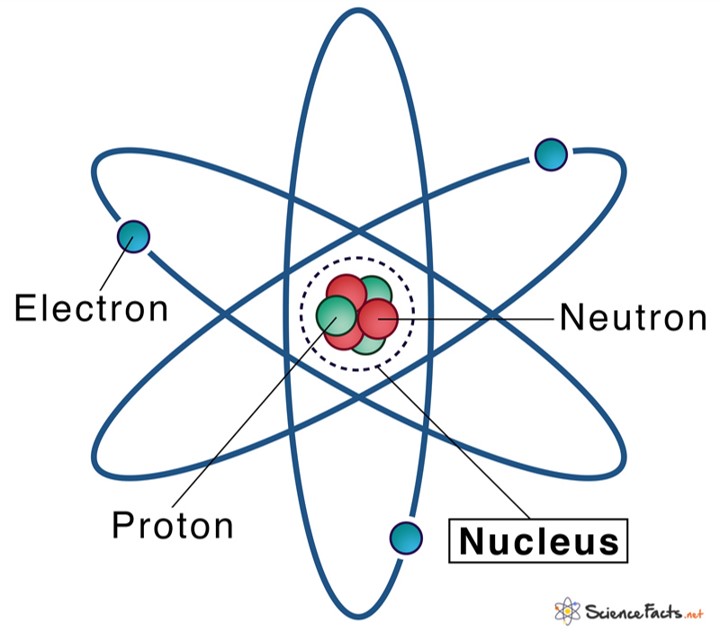
The nucleus (plural, nuclei) is defined as the dense, central part of an atom, consisting of two subatomic particles, namely protons and neutrons. A nucleus accounts for more than 99.9% of an atom’s mass but is 100,000 times smaller than it in size. The word ‘nucleus’ means ‘kernel of a nut’.
Charge of the nucleus #
An atomic nucleus is made of only protons and neutrons. Since protons carry a positive charge (+1) and neutrons are electrically neutral, the nucleus, on the whole, has a net positive charge.
Mass of nucleus #
Proton and neutron have considerable mass, on the other hand, electrons being a fundamental particle it has a legible mass. Thus, the sum of the mass of proton and neutron determines the mass of the atom. In other words, the nucleus contains virtually all the mass of an atom.
Regular units of mass, such as gram (g) or Kilogram (Kg), cannot weigh anything as small as an atom. Thus, scientists have created a new unit of mass. It is called the Atomic Mass Unit (u). Carbon-12 is taken as its reference, and one amu is equal to 1/12th the weight of one atom of Carbon 12.
1 amu = 1.660539 10-24 g