L-isomer #
Because mass spectrometric procedures using nonchiral derivatives and columns do not differentiate the D (+) or L (-) isomers of amphetamine and methamphetamine, it is necessary to perform isomer resolution to determine that a positive result is due to the presence of the D-isomer. Primary examples are the presence of L-methamphetamine in Vicks inhaler, which cannot be distinguished from the use of illicit methamphetamine (D-isomer or racemic mixture depending on method of production) and the excretion of L-methamphetamine and L-amphetamine by patients taking selegiline for Parkinson’s disease. Isomer resolution can be accomplished using a chiral, optically active column, or chiral derivatizing reagents [9]. Use of chiral derivatizing reagents allows analysis on instrument/column systems used for other routine analyses but has the potential disadvantage of possibly obtaining four isomers instead of two if the derivatizing agent is not optically pure. The generally accepted interpretation of isomer resolution results is that >80% of the L-isomer is considered consistent with the use of legitimate medication or conversely >20% of the D-isomer (and total concentration above the cutoff) is considered evidence of illicit use [8,18].
Amphetamine and methamphetamine have optical isomers designated D (or +) for dextrorotatory and L (or -) for levorotatory and the D-isomers, the more physiologically active compounds, are the intended targets of immunoassays. Ingestion of medications containing the L-isomer can cause false-positive results. For example, Vicks inhaler contains the active ingredient L-methamphetamine [17] and extensive use of this product may cause false-positive results for immunoassay screening. Specific isomer resolution procedures must be performed to differentiate the D- and L-isomers.
The dextromethamphetamine (d-isomer) form is highly abused and typically found in illicit preparations.
Reference: [Restek: Analysis of Amphetamines by LC-MS/MS for High-Throughput Urine Drug Testing Labs]
Derivatives #
Reference: Woźniak, M. K., Wiergowski, M., Aszyk, J., Kubica, P., Namieśnik, J., & Biziuk, M. (2018). Application of gas chromatography–tandem mass spectrometry for the determination of amphetamine-type stimulants in blood and urine. Journal of Pharmaceutical and Biomedical Analysis, 148, 58–64. doi:10.1016/j.jpba.2017.09.020
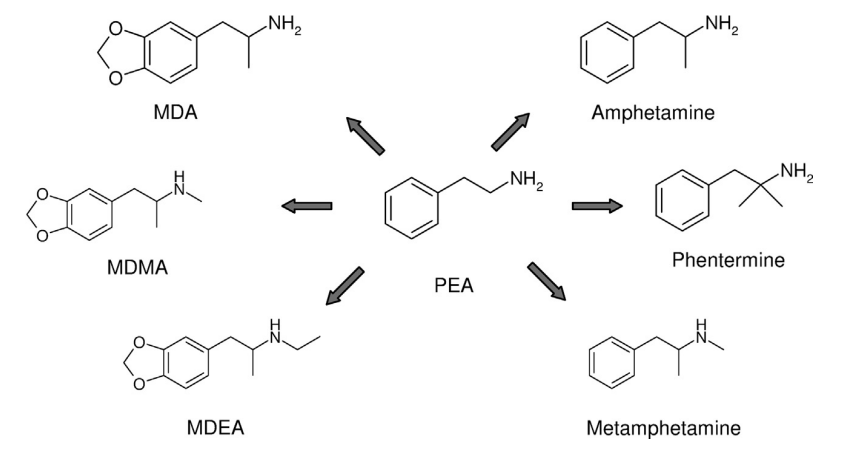
The DEA/FDA to classify amphetamine & methamphetamine as Schedule II controlled substances. Phentermine is a schedule IV drug that is not heavily
abused but is known to exert effects that are similar to Amphetamine.
Designer drugs metylenedioxymethamphetamine (MDMA) and methylenedioxyamphetamine (MDA) are methylenedioxy derivatives of methamphetamine and amphetamine respectively. UCT: AMPHETAMINES IN BLOOD, PLASMA/SERUM, URINE, OR TISSUE USING CLEAN SCREEN® DAU SPE AND LC-MS/MS ANALYSIS
List of interference compounds #
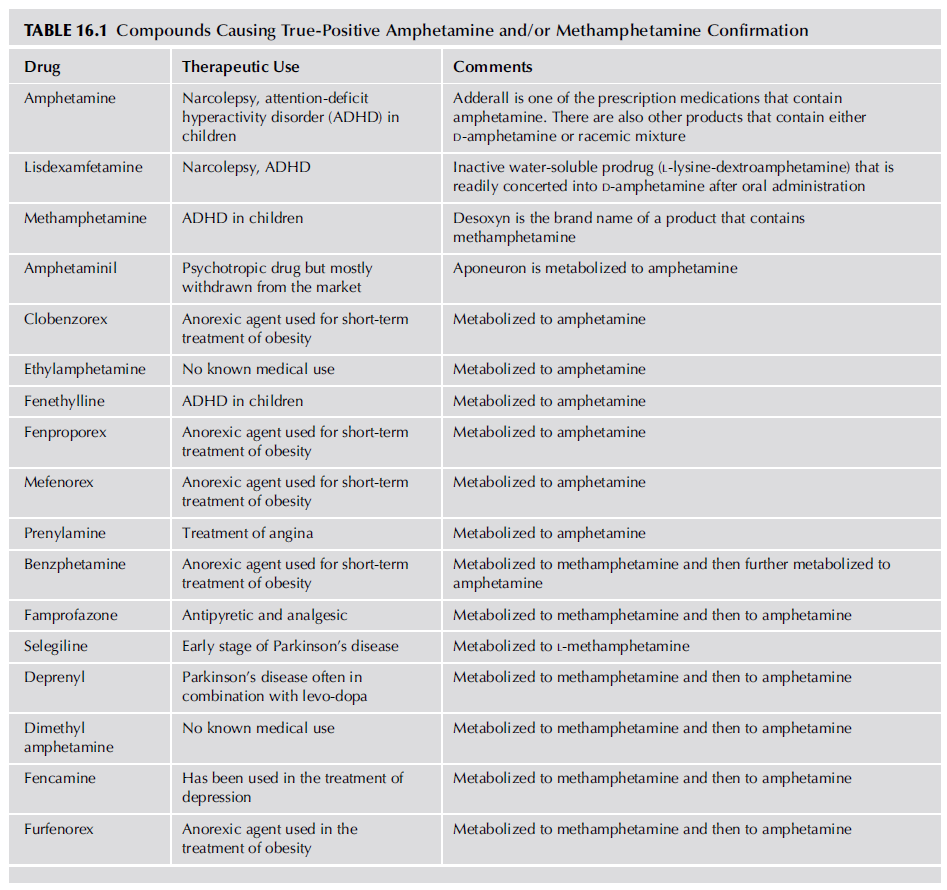
Positive amphetamines results may be also be observed due to ingestion of medications containing amphetamine, methamphetamine, or compounds metabolized to these compounds. Amphetamine [Dexedrine (D-amphetamine), Adderall (D- and L-amphetamine), etc.] and methamphetamine [Desoxyn (D-methamphetamine)] are the active compounds of medications prescribed for appetite suppression, narcolepsy, and attention-deficit disorder and ingestion of these drugs will result in true-positive results due to excretion of these compounds in the urine. Table 16.1 lists compounds whose ingestion can cause positive results due to metabolism to amphetamine and/or methamphetamine.
Fenproporex #
The Federal Drug Administration (FDA) warned consumers in January 2006 that the Brazilian dietary supplements Emagrece Sim and Herbathin contain several active drug ingredients including fenproporex, which is metabolized to amphetamine. Use of other weight loss or nutritional supplements containing fenproporex has resulted in detection of D-amphetamine in the urines of users.
Gabapentin #
Introduction
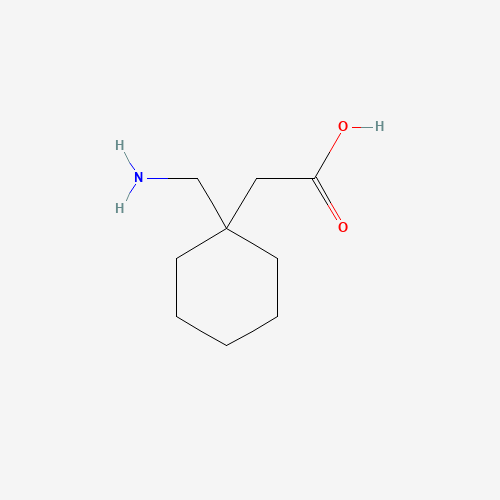
An amphetamine interference was observed during the development of an liquid chromatography–high-resolution mass spectrometry (LC–HRMS) multi-class confirmation method for the determination of 47 drugs and metabolites in urine. The interference passed all qualitative criteria for amphetamine leading to potential false-positive results. Upon investigation, it was found that the amphetamine interference was correlated with the presence of high levels of gabapentin. Gabapentin is routinely detected in patient urine specimens at levels in excess of 1 mg/mL as it is widely prescribed at high doses and does not undergo significant metabolism. The source of the interference was identified as a gabapentin in-source fragment isomeric with protonated amphetamine.
Gabapentin Structure
LC-MS Spectral Info

Precursor m/z 172.133
Ionization Mode: Positive
Top 5 Peaks
172.132889 100
154.122528 87.05
137.095108 31.91
173.135452 9.78
109.100273 6.93
Reference:
- [Characterization of an Amphetamine Interference from Gabapentin in an LC–HRMS Method, Journal of Analytical Toxicology, Volume 44, Issue 1, January 2020, Pages 36–40, https://doi.org/10.1093/jat/bkz046]
- PubChem: Gabapentin