Regular C18 column with LC-MS/MS #
Restek: Analysis of Amphetamines by LC-MS/MS for High-Throughput Urine Drug Testing
Accurate analysis of amphetamines by LC-MS/MS can be achieved using chiral columns, but the analysis presented here provides a simpler, more cost-effective approach for high-throughput labs. Excellent separation of all enantiomers in urine was obtained in a fast, 7-min quantitative analysis on a Raptor C18 column with no interference from matrix components.
Introduction #
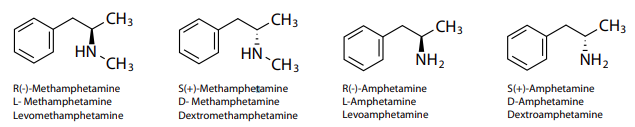
Amphetamine and methamphetamine are psychostimulant drugs that occur as two enantiomers, dextrorotary and levorotary, as a result of their chiral center. The dextromethamphetamine (d-isomer) form is highly abused and typically found in illicit preparations. However, detection of abuse is complicated because consumption of over-the-counter and prescription medications may yield positive results if the analytical method used cannot distinguish between the d- and l- enantiomers. Chiral separation of d- and l-methamphetamine and their metabolites d- and l-amphetamine (Figure 1) can help determine whether the source was licit or illicit, however, chiral columns can be expensive, may necessitate a dedicated instrument, and are not as broadly useful as ubiquitous C18 columns.
In order to provide labs with a high-throughput assay that effectively separates amphetamines by LC-MS/MS in urine without the use of a costly and specialized chiral column, we developed the following analysis on a standard reversed-phase Raptor C18 column. The method employs a simple precolumn derivatization followed by dilution and results in a selective, specific analysis of d- and l-amphetamine and methamphetamine enantiomers in urine that is free from sample matrix interferences.
Liquid-Liquid Extraction #
The trick is to use Marfey’s reagent, 1-fluoro-2-4-dinitrophenyl-5-L-alanine amide.
Analyte-free pooled human urine (BioIVT) was fortified with d- and l-amphetamines and d- and l-methamphetamines (Cerilliant) to prepare seven calibration standards and four QC samples
50 μL of calibration standard or QC sample was aliquoted into a microcentrifuge tube. 10 μL of a working internal standard (20 μg/mL (±)-amphetamine-D11 and (±)-methamphetamine-D11 in water) and 20 μL of 1M NaHCO3 was added and vortexed at 3000 rpm for 10 seconds. After vortexing, 100 μL of 0.1% (w/v) Marfey’s reagent (1-fluoro-2-4-dinitrophenyl-5-L-alanine amide) prepared in acetone was added, vortexed, and heated at 45 °C for 1 hour. Samples were allowed to cool to room temperature before the addition of 40 μL of 1M HCl in water. The sample was then vortexed and evaporated to dryness under nitrogen at 45 °C. Samples were reconstituted in 1 mL of 40:60 water:methanol (v/v) and filtered using Thomson SINGLE StEP standard filter vials (cat.# 25893) and then injected.
Reference #
[Restek: Analysis of Amphetamines by LC-MS/MS for High-Throughput Urine Drug Testing Labs]
Appendix #
Chiral isomer: optical isomer.
Marfey’s reagent, 1-fluoro-2,4-dinitrophenyl-5-L-alanine amide (FDAA), a pre-column derivatizing reagent, is used for the separation of enantiomeric isomers of amino acids and amine compounds. [B’Hymer, C., Montes-Bayon, M., & Caruso, J. A. (2003). Marfey’s reagent: Past, present, and future uses of 1-fluoro-2,4-dinitrophenyl-5-L-alanine amide. Journal of Separation Science, 26(1-2), 7–19. doi:10.1002/jssc.200390019]
Sciex LC-MS/MS chiral column #
Reference: Sciex: LC-MS/MS Chiral Separation of “d” and “l” Enantiomers of Amphetamine and Methamphetamine