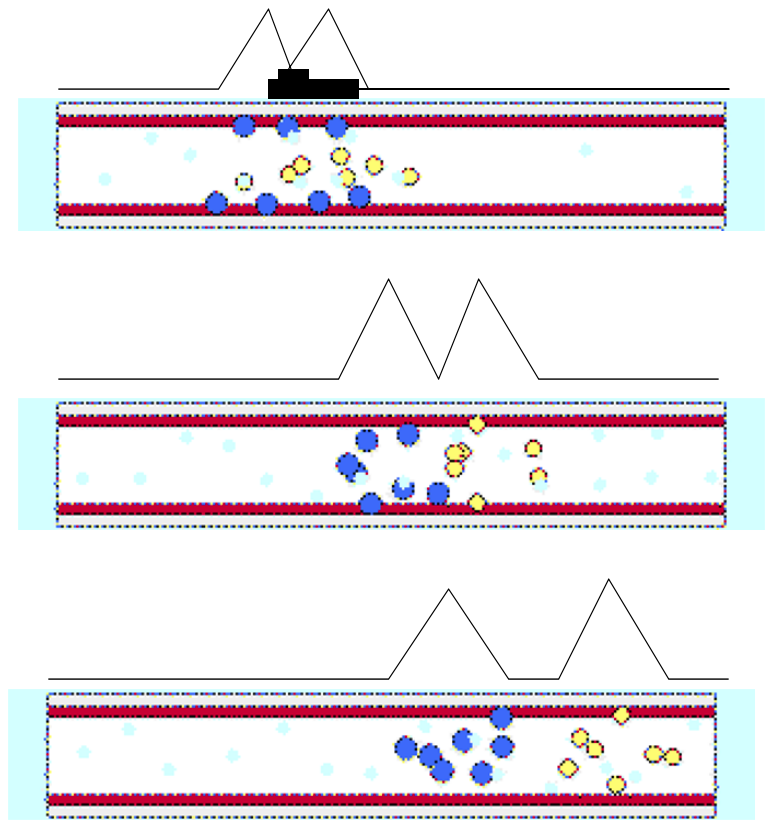
Van Deemter Plot explains resolution.
Resolution is a measurement of how well two species have been separated.
It is useful to relate the resolution to the number of plates in the column, the selectivity factor and the retention factors of the two solute.
To obtain high resolution, the three terms must be maximized.
- An increase in the number of theoretical plates, N, by lengthening the column leads to an increase in retention time and increased band broadening ‐ which may not be desirable. Instead, to increase the number of plates, the height equivalent to a theoretical plate can be reduced by reducing the size of the stationary phase particles.
- It is often found that by controlling the capacity factor, k', separations can be greatly improved. This can be achieved by changing the temperature (in Gas Chromatography) or the composition of the mobile phase (in Liquid Chromatography).
- The selectivity factor, $\alpha$, can also be manipulated to improve separations. When $\alpha$ is close to unity, optimizing k' and increasing N is not sufficient to give good separation in a reasonable time. In these cases, k' is optimized first, and then $\alpha$ is increased by one of the following procedures:
– Changing mobile phase composition
– Changing column temperature – Changing composition of stationary phase
– Using special chemical effects (such as incorporating a species which complexes with one of the solutes into the stationary phase)