Reversed Phase Chromatography(RPC)
In this technique, one uses hydrophobic interactions between the sample and the ligand on the chromatographic support to obtain separation. For proteins, mobile phase additives, such as trifluoroacetic acid, increase hydrophobicity by forming ion pairs that strongly adsorb to the stationary phase. Adsorption is so strong that a gradient of, increasing concentration of organic solvent such as acetonitrile or 2-propanol, is required for elution.
Because of the high ligand density of RPC media and the drastic elution conditions required, the enzymatic and immunologic activity of proteins is generally not maintained after RPC separation. RPC is mainly used for separating small molecules and peptides and is not commonly used for proteins.
The advantage of RPC is that this technique is perhaps the most efficient of all HPLC separation modes. RPC has a high peak capacity and is particularly effective for separating small molecules, peptides, nucleotides, and fragments.
Reversed Phase Chromatography(RPC)
In this technique, one uses hydrophobic interactions between the sample and the ligand on the chromatographic support to obtain separation. For proteins, mobile phase additives, such as trifluoroacetic acid, increase hydrophobicity by forming ion pairs that strongly adsorb to the stationary phase. Adsorption is so strong that a gradient of, increasing concentration of organic solvent such as acetonitrile or 2-propanol, is required for elution.
Because of the high ligand density of RPC media and the drastic elution conditions required, the enzymatic and immunologic activity of proteins is generally not maintained after RPC separation. RPC is mainly used for separating small molecules and peptides and is not commonly used for proteins.
The advantage of RPC is that this technique is perhaps the most efficient of all HPLC separation modes. RPC has a high peak capacity and is particularly effective for separating small molecules, peptides, nucleotides, and fragments. | 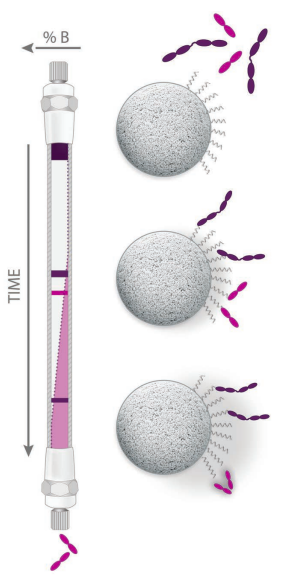
|
Hydrophilic Interaction Chromatography (HILIC)
HILIC is used for the separation of polar and hydrophilic compounds. HILIC stationary phases are polar, similar to normal phase chromatography (NPC), but mobile phases are similar to reversed phase chromatography (RPC). Typical HILIC phases are silica or polymer particles carrying polar functional groups, e.g. hydroxyl, carbamoyl, amino or zwitterionic groups. Typical mobile phases are aqueous buffers with organic modifyers - primarily acetonitrile - applied in isocratic or gradient mode. In HILIC water has the highest elution power. Therefore gradients start with high percentage of acetonitrile.
It is commonly believed that in HILIC the aqueous content of the mobile phase creates a water rich layer on the surface of the stationary phase. This allows partitioning of solutes between the more organic mobile phase and the aqueous layer. Hydrogen bonding and dipole-dipole interactions are supposed to be the dominating retention mechanisms. The number of polar groups, as well as the conformation and solubility of the sample in the mobile phase determine the elution order. Compared to RPC the elution order in HILIC mode is inversed for most compounds.
HILIC is ideally suited for mass spectrometric analysis of water soluble polar compounds, because the high organic content in the mobile phase increases MS detection sensitivity | 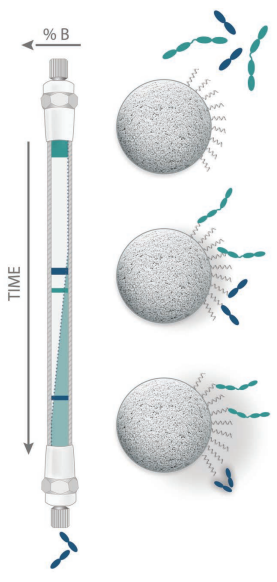
|
Hydrophobic Interaction Chromatography (HIC)
This is a chromatographic technique in which the sample interacts, at high mobile phase salt concentration, with a hydrophobic stationary phase. Subsequently it is eluted from the stationary phase by decreasing the salt concentration. Almost all biological molecules have in their structure hydrophobic patches that, under physiological conditions, are shielded by hydrophilic or ionic groups. By increasing the salt concentration of the solvent, these hydrophobic patches of the molecule become more exposed and can interact with hydrophobic ligands on the HIC packing. HIC is particularly attractive for protein purification when the sample is solved in high salt concentration.
In contrast to the conditions used in RPC, the biological activity of the eluted molecules is often maintained in HIC. It is being used increasingly as a substitute for ammonium sulfate precipitation because of higher throughput and greater recovery of enzymatic activity.
The strength of the hydrophobic interaction is influenced strongly by the nature of the salt components in the mobile phase. Starting salt concentration of 1.0 M to 2.5 M ammonium sulfate in the buffer is commonly used to adsorb the sample to the packing. The salt concentration needed depends on the protein hydrophobicity and solubility, the resins hydrophobicity and the resolution, capacity and mass recovery required. Additives commonly used are methanol, ethanol, isopropanol, acetone, SDS, urea and guanidinium hydrochloride. | 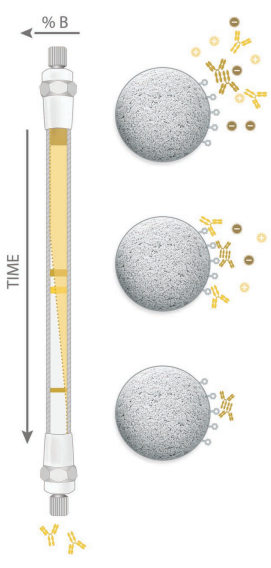
|
Affinity Chromatography (AFC)
AFC is based on the specific adsorption of a molecule to a ligand or
macromolecule. Almost all biological molecules can be purified on the basis of
specific interaction between their chemical or biological structure and a
suitable affinity ligand. Typical molecular pairs are antigens and antibodies,
enzymes and coenzymes, and sugars with lectins.
Affinity Chromatography media have ligands that are bonded via a spacer arm to
the packing material. A specific biological molecule is then reversibly
adsorbed to the ligand. The adsorbed molecule is eluted either by competitive
displacement or by a change in the conformation of the molecule through a
change in pH or ionic strength.
Because of the intrinsic high selectivity of Affinity Chromatography, it is, in
contrast to other chromatographic methods, most suitable for specific
separation problems and provides high purification yields. Another advantage of
AFC is the simplicity of the elution technique, which involves a single-step gradient.
Antibody Affinity columns are used for specific analyses in antibody
therapeutics development, such as fast screening of cell lines for antibody
titer or activity | 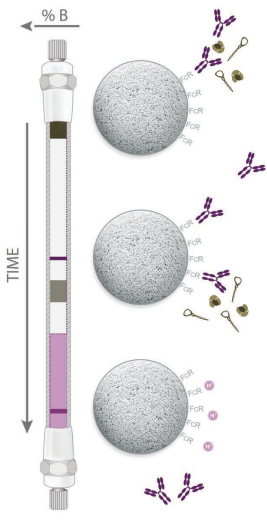
|
Size Exclusion Chromatography (SEC)
SEC is the general name for the chromatographic mode also referred to as gel
permeation chromatography (GPC) for non-aqueous elution systems or gel
filtration chromatography (GFC) for aqueous systems.
SEC is a method in which components of a mixture are separated according to
their molecular size (hydrodynamic volume), based on the flow of the sample
through a porous packing. Large biomolecules that cannot penetrate the pores of
the packing material elute first. These large biomolecules are said to be
excluded from the packing; they flow with the mobile phase in the interparticle
space of the packed column. Smaller molecules can partially or completely enter
the stationary phase. Because these smaller molecules have to flow through
both, the interparticle space, as well as through the pore volume, they will
elute from the column after the excluded sample components.
SEC is a very simple method for separating biomolecules, because it is not
necessary to change the composition of the mobile phase during elution.
However, the separation capacity of this method is limited. For a baseline
separation it is necessary that the molecular weights of the biomolecules
differ at least 10 to 20 %. | 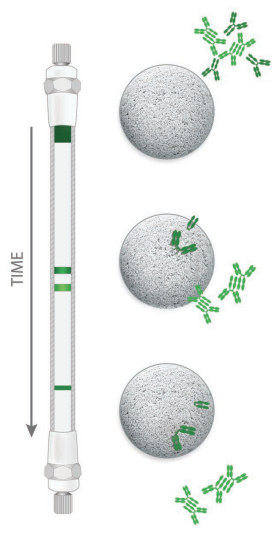
|
Ion Exchange Chromatography (IEC)
Biomolecules generally have charged groups on their surfaces, which change with
the pH of the solution. This is the basis for Ion Exchange Chromatography
(IEC), in which the molecule reversibly binds to an oppositely charged group of
the packing material.
Molecules with a higher charge density bind more strongly to the packing. The
bound sample may be selectively removed from the stationary phase by changing
the pH or salt concentration of the mobile phase. The higher the charge of the
molecule and the stronger the binding to the stationary phase, the greater is
the change in the salt concentration required. In IEC it is possible to load
samples in a very dilute solution and to elute rapidly with a step gradient,
thus producing a concentrated sample.
IEC is a very powerful separation tool because it is highly selective and
specific and has a high capacity. Although the technique is used for a variety
of samples, it is particularly effective for proteins because they are
amphoteric. It is estimated that 70 % of all separation methods for proteins
involve IEC. | 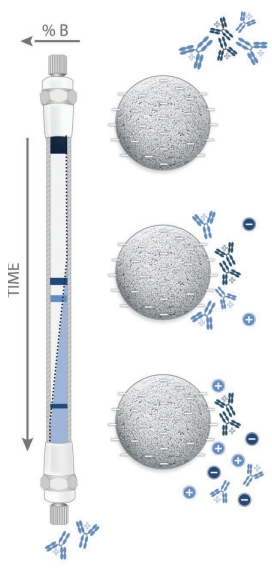
|
Resource Tosoh





