Fundamentals of Mass Spectrometry provided by Waters (2 of 7)
Adduct ions are formed by the interaction of a precursor ion with one or more atoms or molecules, such as hydrogen ions, sodium ions, potassium ions, ammonium ions, hydride ions, chloride ions, bromide ions, acetate ions to form an ion.
Adding particular cations or anions into solution can enhance the formation of adduct ions, such as formic acid, acetic acid.
Matrix suppression is the result of adduct ion formation with background ions.
Compounds that readily form particular cations or anions can be added to sample solutions in order to enhance the formation of adduct ions.
This is a transcript of the following video.
Hello, my name is Helen, I work at Waters Corporation as an analytical scientist. So I’m going to give a short presentation about adduct ions.
As we saw on the electrospray ionization video, the generation of ions in ESI usually involves the formation of adduct ions. Other the ionization techniques, such as atmospheric pressure chemical ionization (APCI), atmospheric pressure photo ionization (APPI), and Matrix Assisted Laser Desorption/Ionization, commonly known as MALDI, can also produce adduct ions.
What is Adduct Ion? #
 "Adduct ions are formed by the interaction of a precursor ion with one or more atoms or molecules to form an ion containing all of the constituent atoms of the precursor ion as well as the additional atoms from the Associated atoms or molecules". Well, that's the formal definition of an adduct ion. But what does that actually mean? Here when we look at the adduct ion formation process in more detail.
"Adduct ions are formed by the interaction of a precursor ion with one or more atoms or molecules to form an ion containing all of the constituent atoms of the precursor ion as well as the additional atoms from the Associated atoms or molecules". Well, that's the formal definition of an adduct ion. But what does that actually mean? Here when we look at the adduct ion formation process in more detail.
Typical Cations and Anions in Formation #
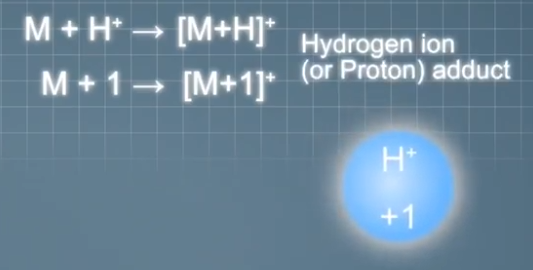
 Typical cations that can form adduct ions in positive mode ESI include hydrogen ions, also known as protons that make singly charged adduct ions one mass unit heavier than the analyte molecules, sodium ions that make the singly charged adduct ions 23 mass units heavier than the analyte molecules,
Typical cations that can form adduct ions in positive mode ESI include hydrogen ions, also known as protons that make singly charged adduct ions one mass unit heavier than the analyte molecules, sodium ions that make the singly charged adduct ions 23 mass units heavier than the analyte molecules,

 potassium ions that make singly charged at adduct ions 39 mass units heavier than the analyte molecules, and ammonium ions that make singly charged at adduct ion 18 mass units heavier than the analyte molecules.
potassium ions that make singly charged at adduct ions 39 mass units heavier than the analyte molecules, and ammonium ions that make singly charged at adduct ion 18 mass units heavier than the analyte molecules.
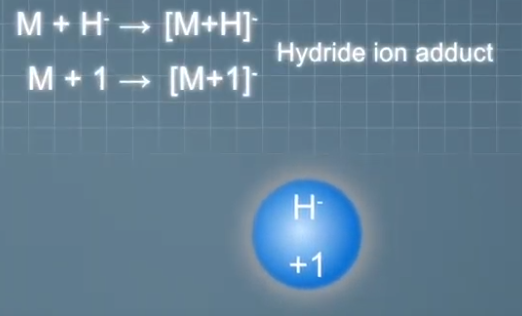
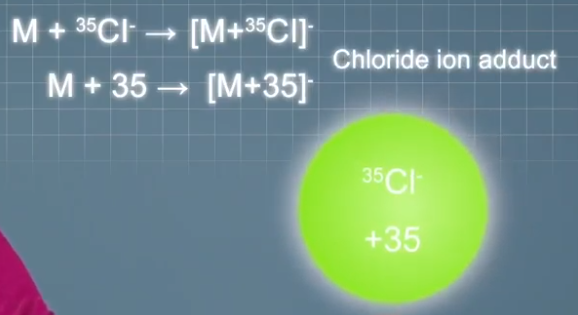 And typical anions that can form adduct ions in negative mode ESI include hydride ions that make singly charged at times were mass unit heavier than the analyte molecules. Chloride ions that make singly charged at times 35 or 37 mass units heavier than the analyte molecules depending on which isotope is forming the adult ion.
And typical anions that can form adduct ions in negative mode ESI include hydride ions that make singly charged at times were mass unit heavier than the analyte molecules. Chloride ions that make singly charged at times 35 or 37 mass units heavier than the analyte molecules depending on which isotope is forming the adult ion.
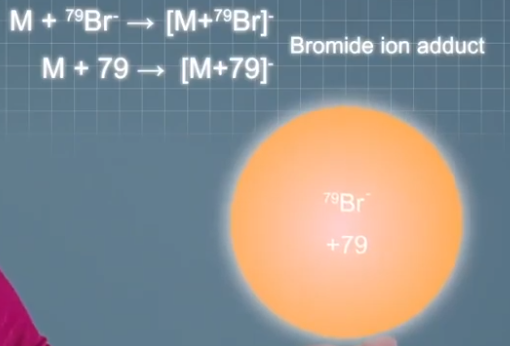
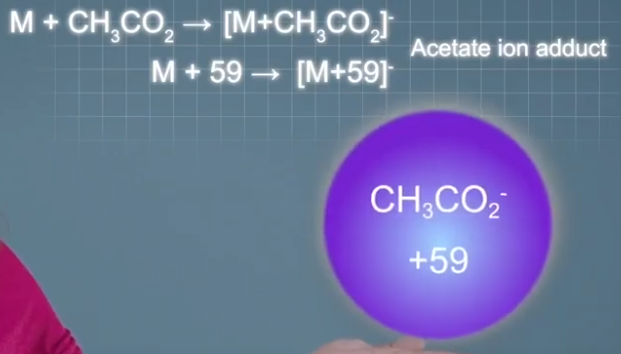 bromide ions that make singly charged adduct ions 79 or 81 mass units heavier than the analyte molecules depending on which isotope is forming the adult ion. And acetate ions make singly charged adduct ions 59 mass units heavier than the analyte molecules.
bromide ions that make singly charged adduct ions 79 or 81 mass units heavier than the analyte molecules depending on which isotope is forming the adult ion. And acetate ions make singly charged adduct ions 59 mass units heavier than the analyte molecules.
Enhancement of Formation #
To enhance the formation of adduct ions, compounds that readily form particular cations or anions can be added to sample solutions. This approach is particularly widely used in polymer analysis, when organic acids or salt solutions are added to polymer solutions to ensure that the analyte ionizes efficiently. Examples of compounds that can be used to enhance adduct ion formation in positive mode include formic acid, also known as methanoic acid, TFA, which stands for Trifluoroacetic acid, and acetic acid, also known as ethanoic acid. These examples all provide hydrogen ions to help enhance protonation.
Alternatively, salt solutions containing specific cations can be used for positive ion mode. For example, Sodium Tetrafluoroacetate, or Silver Tetrafluoroacetate. Silver salts are particularly useful for some polymer analyses. For example, the silver cation bonds really easily with the styrene building blocks used to make polystyrene. Silver has two isotopes with nominal masses of 107 and 109, occurring in the ratio of approximately one to one. This means that adducts formed with silver ions have a characteristic isotope pattern in the mass spectrum.
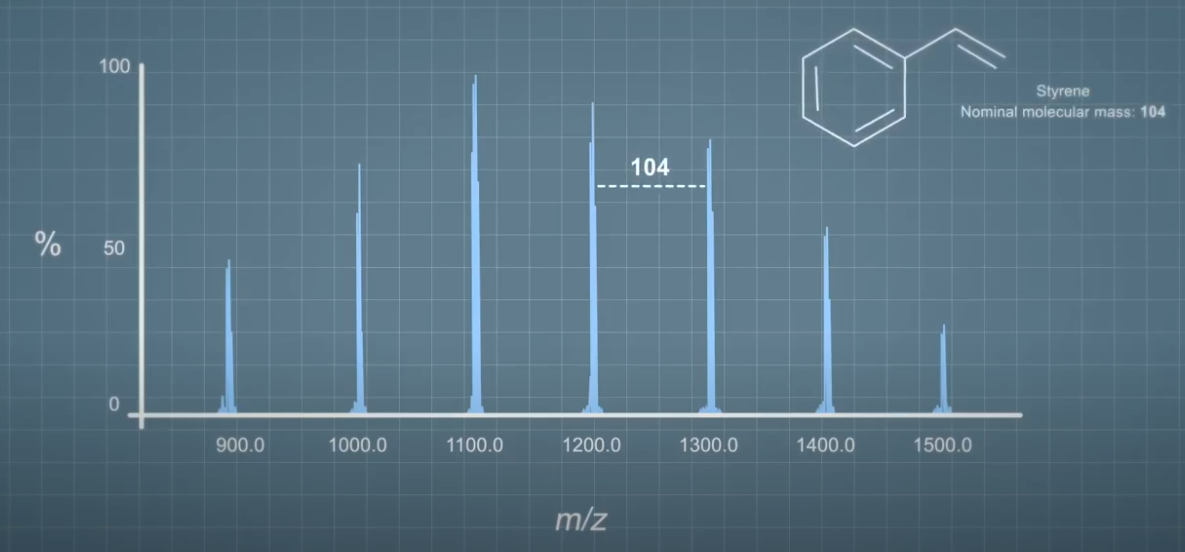
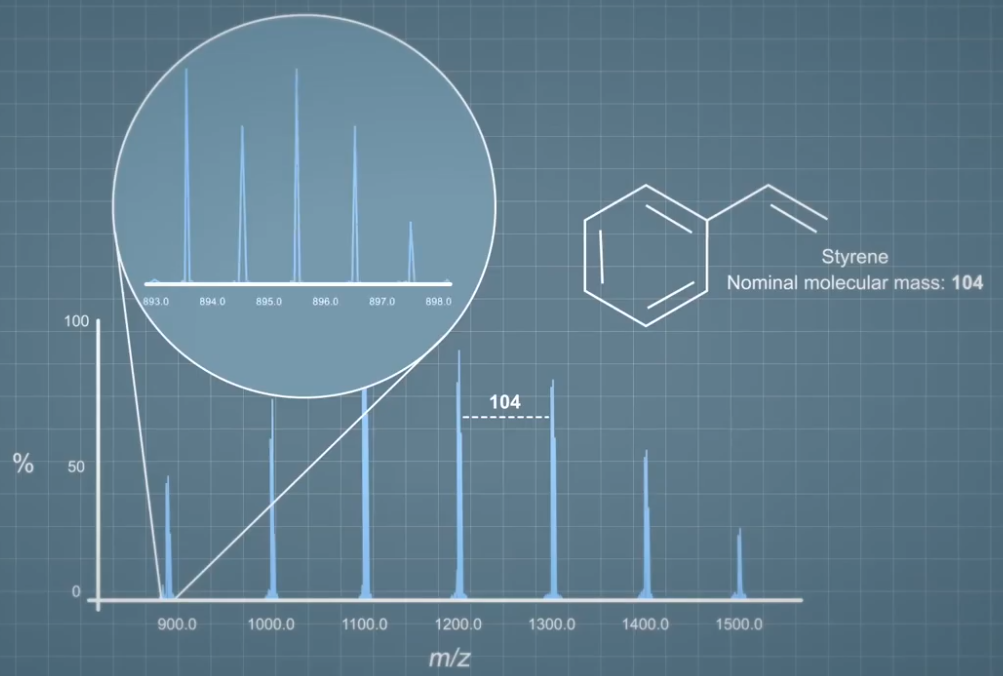 This is a typical mass spectrum of polystyrene, we know this because the gap between each ion cluster in the spectrum is the same as the nominal mass of the styrene molecule that makes up polystyrene. If we take a closer look at one of the ion clusters, we can see that the ions are this unusual pattern which represents the isotope distribution of the ions formed. You can find out more about this in the video covering isotopes.
This is a typical mass spectrum of polystyrene, we know this because the gap between each ion cluster in the spectrum is the same as the nominal mass of the styrene molecule that makes up polystyrene. If we take a closer look at one of the ion clusters, we can see that the ions are this unusual pattern which represents the isotope distribution of the ions formed. You can find out more about this in the video covering isotopes.
Unfortunately, adduct ion formation is not as straightforward as we would like. Sometimes adduct ion formation could be out of our control. For example, older glassware is often a source of unwanted sodium ions. So even if we added a small amount of acid to remote protonation, we might also see some sodium adult ions formed as well.
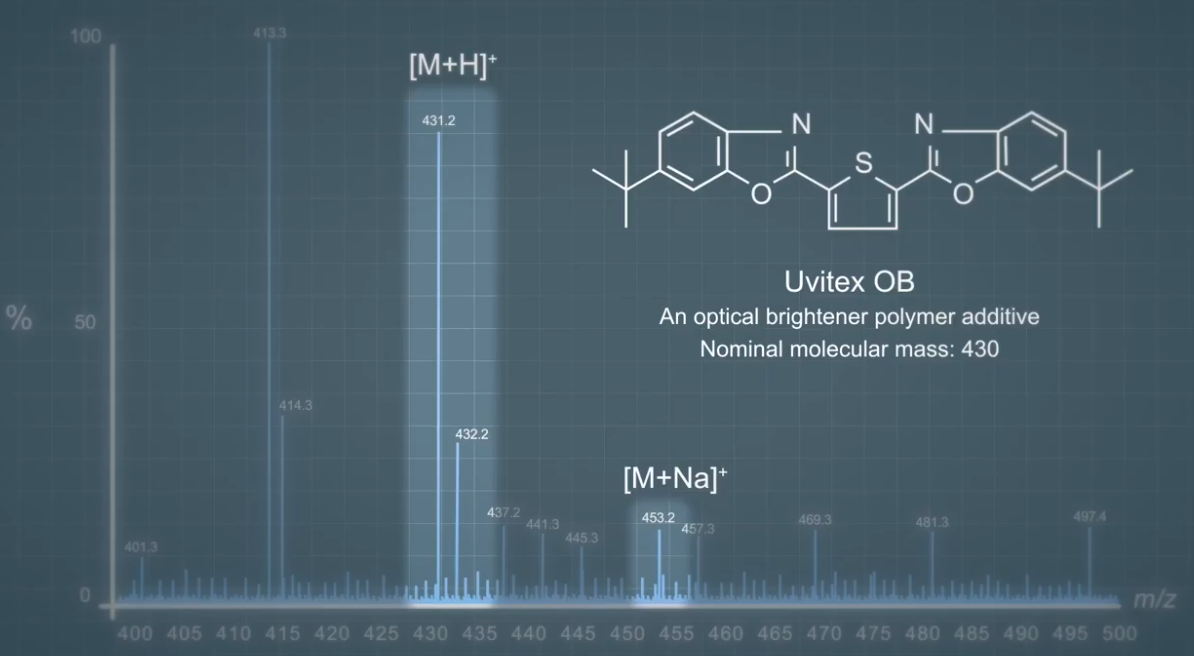 This is a full scan mass spectrum of a sample containing a compound called Uvitex OB, which is an optical brightener polymer additive. This is added to polymers to make them appear and more bright white color. UVitex OB predominantly forms proton adult ions at mass charge 431. And we can see a nice intense peek at that mass charge. We can also see a small amount of sodium adduct ions at 453. When doing an analysis using ESI, we need to look out for the formation of different adduct ions.
This is a full scan mass spectrum of a sample containing a compound called Uvitex OB, which is an optical brightener polymer additive. This is added to polymers to make them appear and more bright white color. UVitex OB predominantly forms proton adult ions at mass charge 431. And we can see a nice intense peek at that mass charge. We can also see a small amount of sodium adduct ions at 453. When doing an analysis using ESI, we need to look out for the formation of different adduct ions.
Deduction of the Site of Protonation #
Knowledge of some simple chemistry principles enables us to deduce the likely site of protonation in analyte molecules. Typically protonation occurs when an atom or atoms in the analyte molecule have at least one lone pair of electrons that are available to form a coordinate covalent bond with a proton. In other words, the cation is most likely to attach to the most basic site on the analyte molecule. Atoms that could be sites where cations might bond include nitrogen, oxygen and sulfur.
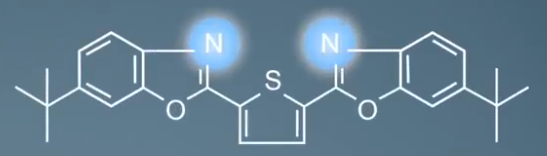 If we take the case of the UVitex OB, we can see that there are several possible sites of protonation, two nitrogen atoms, two oxygen atoms and one sulfur atom. It is likely that the most basic atoms will be the site of protonation. So the proton will probably bond to one of the nitrogen atoms. The space around each atom can also play a part in cation attachment. So, here sulfur is unlikely to bond the proton as it does not have a lot of space around it. We describe it as being sterically hindered.
If we take the case of the UVitex OB, we can see that there are several possible sites of protonation, two nitrogen atoms, two oxygen atoms and one sulfur atom. It is likely that the most basic atoms will be the site of protonation. So the proton will probably bond to one of the nitrogen atoms. The space around each atom can also play a part in cation attachment. So, here sulfur is unlikely to bond the proton as it does not have a lot of space around it. We describe it as being sterically hindered.
Matrix Suppression #
The type of adduct ion formed can also be influenced by the other compounds present with the analyte in the sample mixture. These other compounds are background ions and are known as the matrix. Sometimes matrix components can compete with the analyte’s bond to the cations. If components in the background matrix are more basic than the analyte, then they will form adduct ions at the expense of the analyte of interest. This means that we do not see ions related to the analytes, and we say the analyte response has been suppressed. This is known as matrix suppression.