Atmospheric Pressure Ionization includes ESI and APCI.
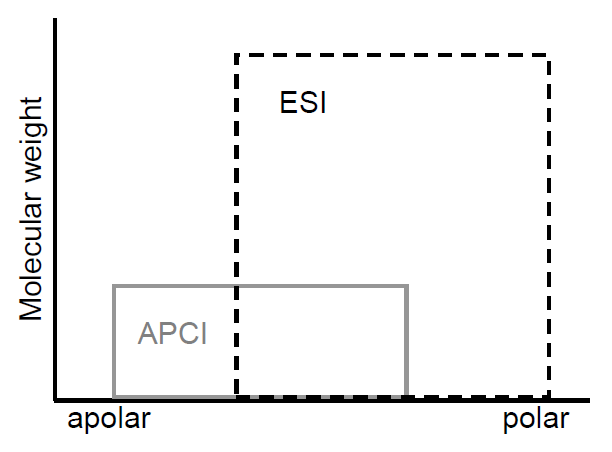
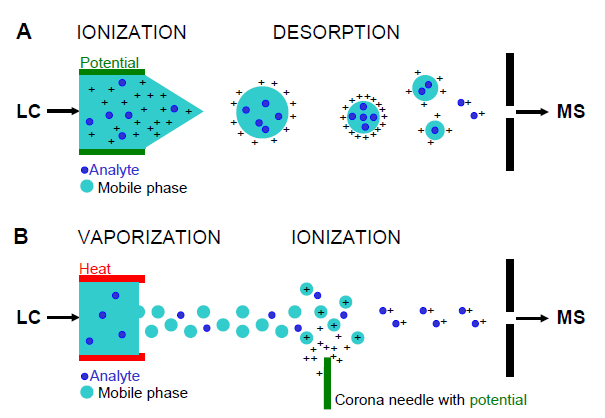
ES and APCI are soft methods of ionization as very little residual energy is retained by the analyte on ionization. The major disadvantage of the techniques is that very little fragmentation is produced. The mass spectra generated by either technique are typically dominated by protonated or deprotonated molecules, [M+H]+ or [M-H]-, depending on the ion mode used and adducts (e.g., [M+Na]+, [M+NH3]+, [M+HCOO]-).
This only provides information on molecular weight. This is very different from the information-rich spectra obtained with EI. For better selectivity through MS/MS or elucidation of structure, fragmentation is needed. Possible fragmentation techniques include “in-source” CID, CID in the collision cell of a tandem-type instrument and fragmentation in an ion trap. One of the major problems encountered using LC-MS with ES is the presence of coeluting matrix compounds that alter the ionization of the target compounds, and which can reduce drastically the response affecting both quantification and detection of pesticide residues. This phenomenon is known as the matrix effect and, because it has an important impact on pesticide analysis, it has been the object of considerable study. There are a number of ways matrix effects can be detected; the most straightforward way is the comparison of the response obtained from a standard solution with that from a standard solution prepared in a matrix extract. This approach can be extended to the comparison of calibration graphs obtained from the analysis of standards prepared in solution with those prepared in matrix extracts. A third approach is the postcolumn infusion system, in which continuous postcolumn infusion of the analyte of interest is performed while blank extracts are injected into the LC column. This enables the evaluation of the absolute matrix effects on the analyte at different portions of the chromatogram, illustrating the need for change in the LC separation required to minimize the matrix effect. If matrix suppression cannot be eliminated by improved sample preparation or reoptimization of LC conditions, careful consideration of calibration strategy is needed to compensate as much as possible for matrix effects. Using matrix-matched calibrants, standard addition or stable isotope-labeled internal standards is recommended.
p78, Analysis of Pesticides in Food and Environmental Samples, CRC Press, 2008, edited by José L. Tadeo
[1] LC-MS/MS in forensix toxicology: What about matrix effects?, Jan, 2011, Ruth Verplaetse
[2] LC-MS/MS in forensix toxicology: What about matrix effects?, Jan, 2011, Ruth Verplaetse