The LC eluate is sprayed into a spray chamber at atmospheric pressure in the presence of a strong electrostatic field and heated drying gas.
The electrostatic field occurs between the nebulizer, which is the ES probe, and the capillary. Aerosol of charged droplet formation at right angles to the capillary.
Ionization takes place as a result of imparting the strong electrical field and the superheated nitrogrn sheath gas, with Agilent AJS (Jet Stream) technology.
Understanding how ions are liberated from the liquid mobile phase in the gas-phase transition helps us understand and diagnose issues such as lack of expected sensitivity and ion suppression.
By Balogh
Two separate theories have been proposed, 1) the charge residue mechanism and 2) the ion evaporation mechanism models, but Cole argues that both mechanisms might be working concurrently: the charge residue mechanism dominating at high mass and ion evaporation dominating for lower masses.
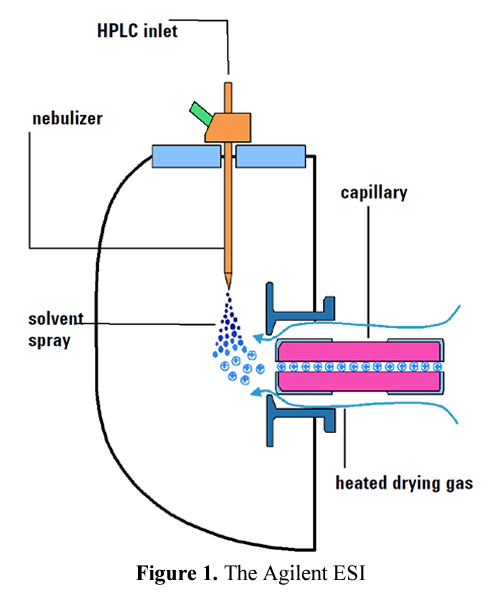
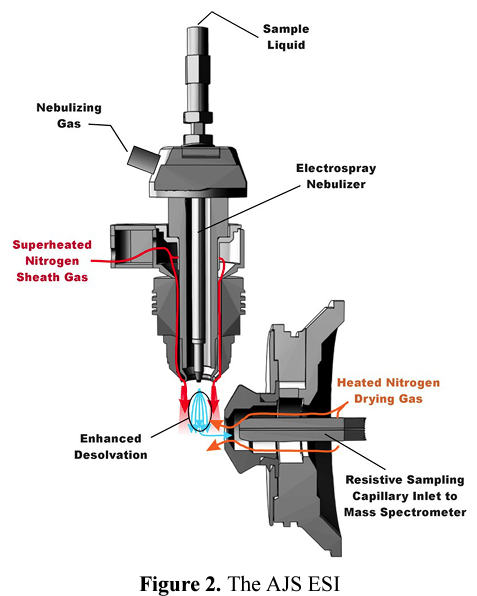 The ES probe, or device, is typically a conductive capillary, usually made of stainless steel, through which the eluent from the LC flows.
The ES probe, or device, is typically a conductive capillary, usually made of stainless steel, through which the eluent from the LC flows.
A voltage is applied between the probe tip and the sampling cone. In most instruments, the voltage is applied on the capillary, while the sampling cone is held at low voltage. The capillary, contained within a larger bore tube, allows a concentric nitrogen flow applied to the aerosol at its exit point so that capillary acts as a nebulizer.
While this variant was initially called “pneumatically assisted electrospray” or “ion spray”,this terminology appears to be replaced by the more generic “electrospray”.
Aerosol droplet formation is enhanced by the added shear forces of the gas and heat transmitted from adjacent supplemental devices, direct heating of the gas itself or with the assistance of an additional heated desolvation gas.
ES ionization takes place as a result of imparting the strong electrical field to the eluent flow as it emerges from the nebulizer, producing an aerosol of charged droplets.
Due to the solvent evaporation, the size of the droplet reduces, and, consequently, the density of charges at the droplet surface increases. The repulsion forces between the charges increase until there is an explosion of the droplet.
These coulombic fissions continue until droplets containing a single analyte ion remain. The charge residue model suggests that a gas-phase ion forms only when solvent from the last droplet evaporates. In the ion evaporation model, the electric field strength at the surface of the droplet is thought to be high enough for solvated ions to attain sufficient charge density to be ejected from the surface of the droplet and transfer directly into the gas phase without evaporation of all the solvent.
Ensuring that the compound of interest is ionized in solution critical for ES ionization, so mobile phases should have a pH such that the analytes will be ionized. Charging is usually accomplished by adding or removing protons but cation or anion attachment generating adduct ions is also common.
p79, Analysis of Pesticides in Food and Environmental Samples, CRC Press, 2008, edited by José L. Tadeo