The LC elute passing through a nebulizing needle, creates a fine spray. The spray passes through a heated ceramic tube, where the droplets are fully vaporized. The resulting gas/vapor mixture is then passed over a corona discharge needle, where the solvent vapor is ionized to create reagent gas ions.
For successful APCI, the analyte must be volatile and thermally stable and the mobile phase must be suitable for gas-phase acid–base reactions.
To vaporize the solvent and analyte, the APCI source is typically operated at vaporizer temperatures of 400 to 500 0C.
APCI enables the analysis of compounds that resisted converting to gas phase ions using ESI, that is, the less polar and more volatile ones.
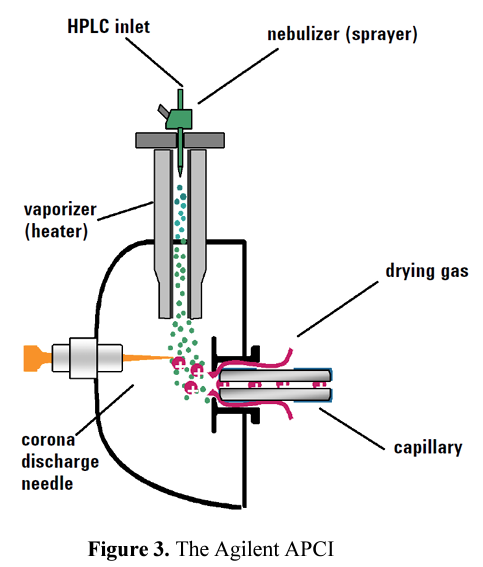 APCI transfers neutral analytes into the gas phase by vaporizing the LC eluent contained within a nonconductive capillary inserted in a coaxial pneumatic nebulizer through which a gas is added to assist the ionization process.
APCI transfers neutral analytes into the gas phase by vaporizing the LC eluent contained within a nonconductive capillary inserted in a coaxial pneumatic nebulizer through which a gas is added to assist the ionization process.
The mixture of gas and nebulized eluent passes through a heated zone that assists the solvent evaporation and the fine droplets are converted into desolvated molecules in the gas phase.
The desolvated analyte molecules are then ionized via chemical ionization; the transfer of charged species between a reagent ion and a target molecule to produce a target ion that can be mass analyzed.
The corona-discharge needle in the APCI source produces a stream of electrons that ionizes the atmosphere surrounding the corona electrode, which consists mainly of nebulizer and drying gases (typically nitrogen and/or air), the vapor generated from the HPLC eluent, and the analyte molecules. The process starts by ionizing nitrogen and finishes with protonated water, water clusters, and solvent clusters as possible reagent ions.
For successful APCI, the analyte must be volatile and thermally stable and the mobile phase must be suitable for gas-phase acid–base reactions. For example, when working in positive ion mode, the proton affinity of the analyte must be higher than the proton affinity of the eluent: that is, the analyte can acquire a proton from the protonated solvent.
Since water cluster ions are a major source of reagent ions, the proton affinity of these clusters relative to analyte ions will have a profound effect on sensitivity. Similarly, the use of certain modifiers added to the mobile phase to enhance LC separation (e.g., triethylamine) can be the source of considerable ion suppression in APCI. A strong base will receive protons from the reactant ions to form their protonated forms. Subsequent proton transfer will occur only if the analyte is more basic than the modifier. Although the choice of the most appropriate interface as well as detection polarity are based on analyte polarity and LC operating conditions, many classes of compounds perform well using either technique and sometimes in both ion modes, whereas, for other compounds, the choice is more restricted. Interfaces are selected based on individual preference derived from experience and available techniques as well as the magnitude of any matrix effects.
Although there are a great number of examples of the use of APCI for pesticide residue analysis for both environmental and food applications, including some pioneering early work, more recently the technique appears to be left in the wake of ES ionization’s overwhelming popularity. This may be related to the increasing number and wider range of pesticides currently sought but perhaps also reflects the improvements in source and probe design for ES not yet paralleled with APCI.
The choice between ES and APCI is irrelevant when using the recently introduced multimode sources, which deliver simultaneous ES ionization and APCI.
p79, Analysis of Pesticides in Food and Environmental Samples, CRC Press, 2008, edited by José L. Tadeo