M+ peak represents the compound. It is often referred to as the parent peak.
M+1 peak represents the compound with isotope. The chance of finding a C-13 is one in 100. C-13 and N-15 can be responsible for M+1 peak.
M+2 peak represents the compound having either chloride or bromide isotope.
Today we’re going to take a look at some more spectroscopy understanding the M+, M+1, and M+2 signals that can sometimes show up on a mass spec. So this is going to usually involve when you’re doing organic spectroscopy, although it’s not limited to that. But this is where this is going to be geared towards as far as this discussion here.
M+ Peak #
Which is the most common peak of the bunch that we’re going to be discussing here, in fact, it’s mandatory, you should always have an M+ peak with your mass spec, it is often referred to as the parent peak, because it is the peak that will represent the compound you’re actually analyzing in the mass spectrometer. So what is the compound? It is only had a single electron removed is representing is the mass of the compound that has been put into the instrument. Now, typically, this peak is going to be clustered in with the highest grouping of peaks in the highest M to Z ratio. So what that means is if we look at m to Z ratio, which is mass to charge, and if it’s just plus, it’s a one, so it’s just referring to the mass here, if you take a look at your mass spec, and you go up to the area where you see the last grouping of peaks, usually the peak that has the highest intensity out of that grouping is going to be the parents peak. Now it doesn’t mean it’s going to be the tallest peak in the entire mass spec, we call that the base peak. If you go up to that region, where you find the highest set of peaks, the one that is the highest out of that group is usually considered the parent peak, which we again referred to as the M+ peak. Alright, now usually what this means is, this peak will represent your individual molecule with the most common set of mass isotopes present. So for instance, if you’re dealing with an organic compound, it will most likely be dealing with a compound that is completely made out of C-12, hydrogen, N-14, O-16. So as I’m listing off these isotopes, these are the most common isotopes. For instance, if I have carbons present, there’s about a 99% chance that it’s a C-12 atom. And so most of the molecules I would expect, that I am injecting into this mass spectrometer are going to be made up of carbon twelves. Every one and 100 might be a C-13. And that leads us into the next topic here, which is the M+1 peak.
M+1 Peak #
So keeping in mind that we’re really talking about the mass to charge ratio, when we say M+1, it represents the fact that our mass because the charge is typically going to be one when we’re using M to Z ratio, the mass is simply one amu, or one Dalton in whatever unit you want to use. It’s one atomic unit heavier. The parent molecule contains a single atom in it, that is going to have an isotope with one mass unit heavier than most of the common isotopes. So again, a C-13, instead of a C-12. The larger the molecule structure, the more likely this peak is to occur and the more prominent the M+1 peak will become. Now this is nothing but a numbers game when you look at it statistically. So if I have a relatively small organic compound, let’s say it only has five carbons that I want to analyze, while the chance of finding a C-13 is one in 100. So there’s going to be a pretty small chance that out of those five carbons, one of them is going to be a C-13. But as I increase in size, so let’s say that now I’m starting to analyze a compound that has 40 carbons. Well, now there’s a much more higher likelihood or a greater likelihood that I could potentially see a C-13. In one of those 40 carbons, since the odds are about one in 100, that a C-13 will randomly occur. Inorganic compounds, as long as you have a somewhat modest size, then you will usually see an M+1 peak. And that is most commonly due to a C-13 isotope. It’s about a one in 100 chance. So you can easily see M+1 peaks in most organic compounds provided, I would say they have at least somewhere between five to seven carbons. Now, nitrogen is another possibility if you have a compound that is rich in nitrogen, because remember, organic compounds are gonna to have far more carbon than nitrogen most often, he but if you do have an organic compound that’s relatively rich in nitrogen, you could potentially see a N-15, that is resulting in a M+1 peak, but this is less likely than the C-13. So if you look at the natural abundance, the nitrogen has about a one in 200 chance of becoming an N-15 instead of the one in 100 chance of the C-13. So again, nitrogen could be responsible for an M+1 peak. But if there’s carbon present, especially if there’s more carbon than there is nitrogen, most of the time, the M+1 peak is going to be due to the fact that there’s C-13 Present. Right now, if you are studying non organic compounds, or potentially, if you’ve got a weird organic compound that has a whole lot of either silicone or sulfur, then you may also see an M+1 peak. In fact, silicone, if I am not mistaken, has about a one in 20 chance, it’s about a 4% for the M+1 isotope of silicone.
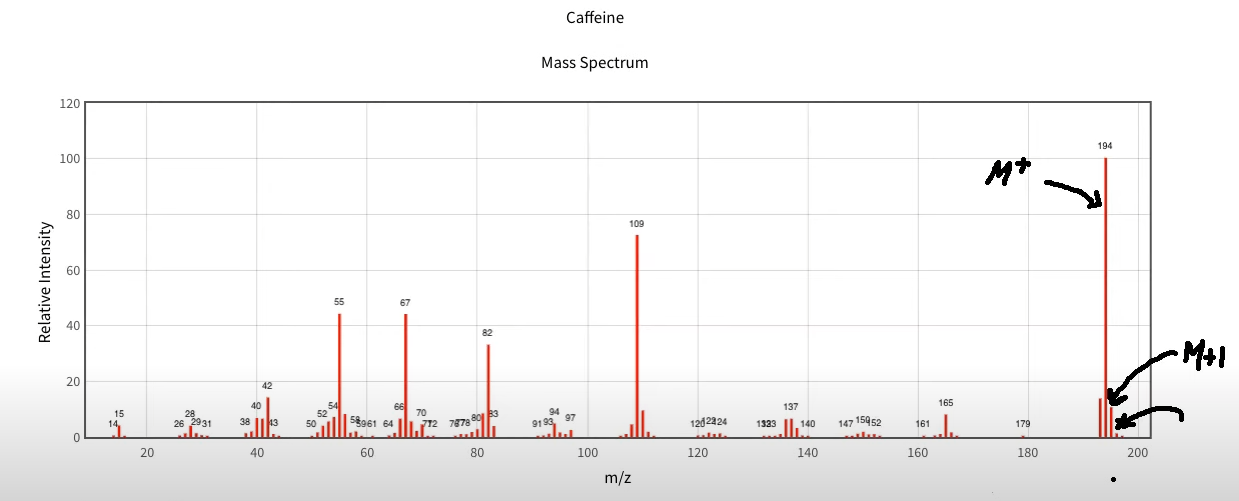 So let's take a look here before we go to M+2, we have caffeine this is the mass spectra for caffeine. And what I want you to notice here is if we come over the grouping of peaks right here, are considered to be the highest group of peaks as far as the `M to Z ratio` is concerned. So of that grouping, we can see one of them is far taller as far as the relative intensity is concerned than the others. And that is this one right here. This is the M+ peak at 194. So that's the M+ for caffeine. And then if you take a look, it's pretty easy to see this here, we have a peak right next to it. Now it's not labeled as 195. But that's what it would be, then this would be the M+1 peak. And then you can also have the M+2 and that is there. It's very tiny relative to the M+1. And we'll see why in a minute. particularly has to do with as you continue to go up the ranks like that, there's less and less chance that you're going to see these types of peaks now that could be due to two C-13. So although it's less likely, you'd have to have a whole lot of them. it's probably due to oxygen because oxygen can give an M+2 peak.
So let's take a look here before we go to M+2, we have caffeine this is the mass spectra for caffeine. And what I want you to notice here is if we come over the grouping of peaks right here, are considered to be the highest group of peaks as far as the `M to Z ratio` is concerned. So of that grouping, we can see one of them is far taller as far as the relative intensity is concerned than the others. And that is this one right here. This is the M+ peak at 194. So that's the M+ for caffeine. And then if you take a look, it's pretty easy to see this here, we have a peak right next to it. Now it's not labeled as 195. But that's what it would be, then this would be the M+1 peak. And then you can also have the M+2 and that is there. It's very tiny relative to the M+1. And we'll see why in a minute. particularly has to do with as you continue to go up the ranks like that, there's less and less chance that you're going to see these types of peaks now that could be due to two C-13. So although it's less likely, you'd have to have a whole lot of them. it's probably due to oxygen because oxygen can give an M+2 peak.
M+2 Peak #
Alright, so the M+2 peak, the two most common occurrences of M+2 by far are due to chlorine and bromine presence in a compound. So these are very obvious even more so than the M+1 C-13 peaks. So chlorine has a set of isotopes, Cl-35, and Cl-37. The 35 is the more common one, and the 37 is less. So it’s about a 75 to 25% mix, or a three to one ratio. Alright, so if chlorine is present, the M+ peak is going to be about three times as intense as your M+2 peak. So if your M+2 peak is about a third the size of your parent peak go up and look at caffeine. If you had one that was about the third the size of this peak, that’s going to be very evident in the M+2 spot, you’ll see a pretty large peak there. now, bromine, you have bromine 79 and bromine 81. They are in a one to one ratio, relatively speaking for isotopes. And so you will see an M+ that has an equal intensity peak at M+2, they will be almost dead even with one another as far as the statistical occurrence of them. so chlorine and bromine are the two most common sources of M+2 peaks, especially ones that are very prominent that we can easily see. Now, if an organic molecule is sufficiently large enough, and it has to be pretty large in order to pull this off, it could potentially have an M+2 peak due to two C-13s in one molecule. Now again, the chance here is one in 100. So now you’re trying to take that one and 100 chance and say it’s going to happen twice in the same molecule. You are getting into shaky territory in terms of statistics, it’s not that it’s impossible, but it starts to become pretty improbable. Unless, you’re going to have a very large organic molecule where you’ve got a whole bunch of carbons. And it’s certainly more possible at that point that you could see several C-13s that might be present. Same thing for a C-13 and a N-15. So we could have one C-13 and one N-15. This would be even less likely because N-15 is harder to get a hold of than C-13. So to have both of them in the same molecule, again, it’s going to be pretty rare, you would have to have a pretty big molecule with a lot of carbon and a lot of nitrogen to pull this off. But it is possible.
The other possibilities that we can have are going to be less likely than the halogens, but they will still be more likely than the C-13 and N-15 examples that we just discussed. So these could include oxygen, we have O-16 and O-18, the silicone and the sulfur because silicon and sulfur both have M+1 and M+2 isomers or isotopes that we can take a look at.
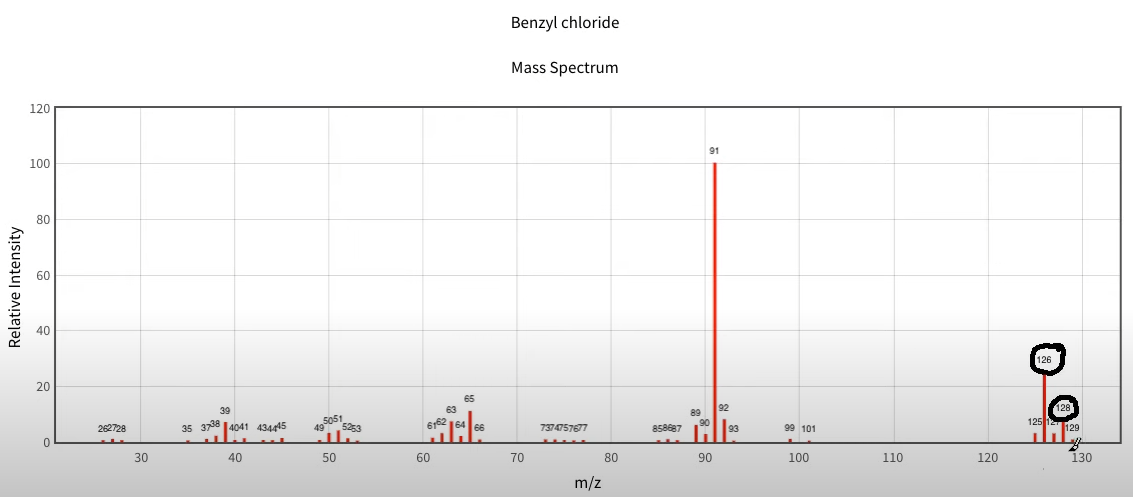
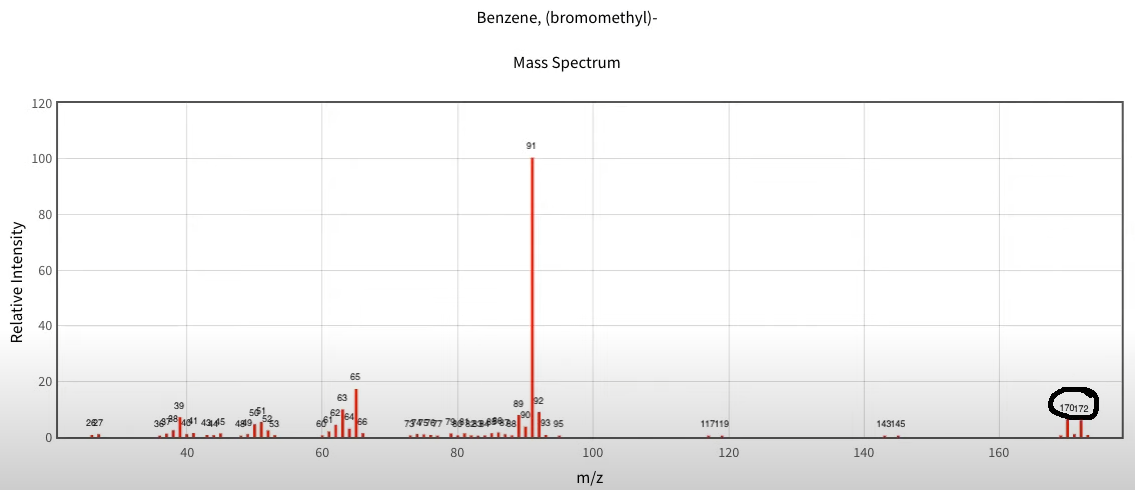 So here's an example with benzyl chloride and then we're going to take a look at benzyl bromide. So you can see here's again, the grouping in the highest `M to Z ratio` area 126 would be the parent peak, because it is most definitely the tallest of the group. And I can see about a third that size is 128. And that would be the chlorine isotopes that are behaving there. So then if I go down, and I take a look at the benzyl bromide, here I see 170 and 172. And look at that they're almost equal in terms of their height to one another. And again, here is the highest group in terms of the `M to Z ratio`.
So here's an example with benzyl chloride and then we're going to take a look at benzyl bromide. So you can see here's again, the grouping in the highest `M to Z ratio` area 126 would be the parent peak, because it is most definitely the tallest of the group. And I can see about a third that size is 128. And that would be the chlorine isotopes that are behaving there. So then if I go down, and I take a look at the benzyl bromide, here I see 170 and 172. And look at that they're almost equal in terms of their height to one another. And again, here is the highest group in terms of the `M to Z ratio`.